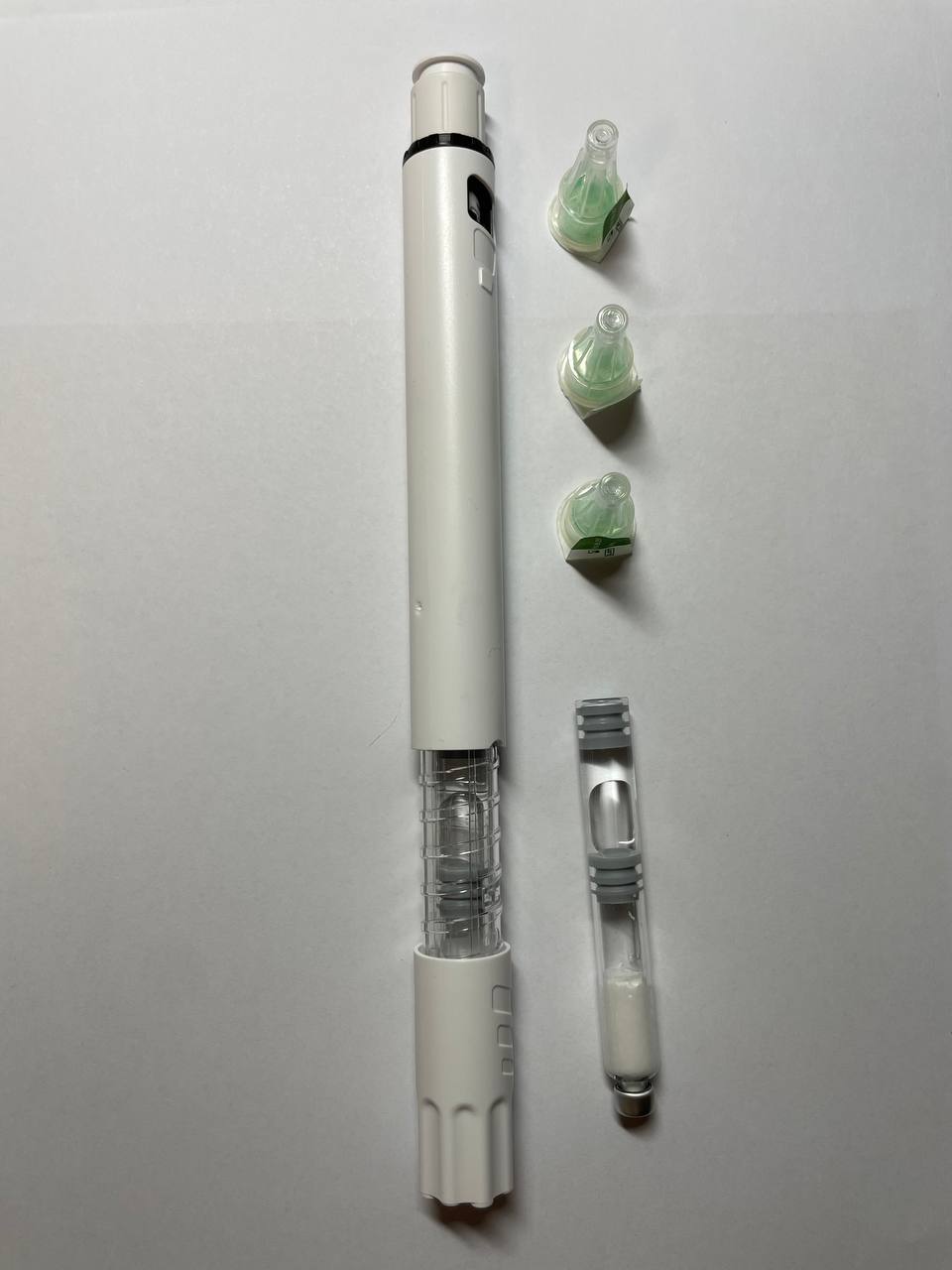
Retatrutide 50mg Dual-Chamber Cartridge Pen
£110.00 Original price was: £110.00.£55.00Current price is: £55.00.
Retatrutide 50mg Dual-Chamber Cartridge Pen
What is Retatrutide?
Retatrutide is an investigational weight‑loss medication in clinical trials. It belongs to the family of incretin mimetics, related to GLP‑1 receptor agonists such as semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound). Unlike current medicines, retatrutide is a triple agonist that targets GLP‑1, GIP, and glucagon receptors—under study for potential effects on weight, non‑alcoholic fatty liver disease (NAFLD), and metabolic health.
How Retatrutide Works (GLP‑1 Medication)
Retatrutide’s three‑receptor approach is designed to act on multiple pathways involved in appetite, glucose control, and energy use:
- GLP‑1 receptor activation: may slow digestion, reduce appetite, and lower blood glucose.
- GIP receptor activation: may improve insulin sensitivity and support fat metabolism.
- Glucagon receptor activation: may increase energy expenditure and fat burning.
This multi‑pathway strategy is what sets retatrutide apart from current GLP‑1‑only medicines.
Retatrutide Injection
- Given as a once‑weekly injection.
- Administered with a pen‑style injector designed for self‑use after proper training.
- Injection sites: thigh, upper arm, or abdomen (rotate sites).
Retatrutide Dosage & Administration (under study)
Clinical studies have explored a gradual dose escalation to improve tolerability. A commonly referenced step‑up schedule under investigation looks like:
| Week | Dose |
|---|---|
| 1–4 | 2 mg |
| 5–8 | 4 mg |
| 9–12 | 8 mg |
| 13+ | 12 mg (maintenance) |
How to Take Retatrutide
- Use the pen injector once per week, ideally on the same day each week.
- Rotate injection sites (thigh, upper arm, abdomen) to reduce skin irritation.
- If you miss a dose, follow your clinician’s advice or the official instructions that come with your medicine. Do not double up a dose without medical guidance.
- Never share your pen with anyone else. Retatrutide 50mg
Retatrutide Side Effects
As with other incretin‑based medicines, side effects are often gastrointestinal—especially at the start or when increasing the dose:
Common
- Nausea
- Vomiting
- Diarrhoea or constipation
- Abdominal pain or cramping
- Reduced appetite
Less common but potentially serious (being studied)
- Gallbladder problems
- Pancreatitis
- Possible thyroid effects (seen with some GLP‑1s in animal studies)
Retatrutide Pen & Instructions
- Remove the cap.
- Select and clean the injection site (rotate weekly).
- Press the pen against the skin and press the button/click to release.
- Hold in place for the full injection time per the instructions you were given.
- Place used pens/needles in a sharps container—do not discard in household waste.
When approved products become available (if applicable), expect step‑by‑step training and clear patient instructions similar to other GLP‑1/GIP medicines.
FAQs
Is retatrutide approved?
Retatrutide is currently being investigated in clinical trials. Regulatory status can change—your clinician or local medicines regulator will have the latest information.
How does retatrutide compare to semaglutide or tirzepatide?
They work on overlapping—but not identical—targets. Retatrutide is being studied as a triple agonist (GLP‑1/GIP/glucagon), while semaglutide is GLP‑1–only and tirzepatide is a dual GLP‑1/GIP agonist. Head‑to‑head clinical outcome data and official guidance are needed for robust comparisons.
What should I do if I miss a dose?
Follow the specific instructions provided by your clinician or the official patient leaflet. As a general safety principle, avoid double‑dosing. If you’re unsure what to do, seek medical advice.
Brand names mentioned—Ozempic, Wegovy, Mounjaro, Zepbound—are trademarks of their respective owners and are used here for educational context only. Retatrutide 50mg

Reviews
There are no reviews yet.